
Small Molecules Activation
Various sterically hindered ligands will be synthesized and their co-ordination chemistry will be explored to stabilize unusual oxidation states and coordinatively unsaturated metal complexes. Their catalytic activity against small molecule such as CO2, N2, O2 etc. will be probed in details to convert them into useful small organic molecules. Detailed mechanistic studies will be investigated to fine tune the catalytic activity
Our research focuses on the design and development of transition metal–based catalysts for small molecule activation and energy-relevant transformations. By tailoring the coordination environment around 3d transition metals, we aim to control reactivity, selectivity, and efficiency in catalytic processes that are central to sustainable chemistry and energy conversion.
A major thrust of our work lies in catalytic transformations such as CO₂ reduction (CO₂RR), hydrogen evolution reaction (HER), oxygen reduction reaction (ORR), and water oxidation, along with overall water splitting. These reactions are fundamentally important for addressing global challenges related to carbon utilization and clean energy generation. CO₂ reduction provides pathways for converting greenhouse gases into value-added chemicals and fuels, while HER and ORR are key electrochemical processes in fuel cells and electrolyzers. Water oxidation and overall water splitting play a crucial role in artificial photosynthesis and hydrogen production, offering sustainable routes to generate molecular oxygen and hydrogen fuel.
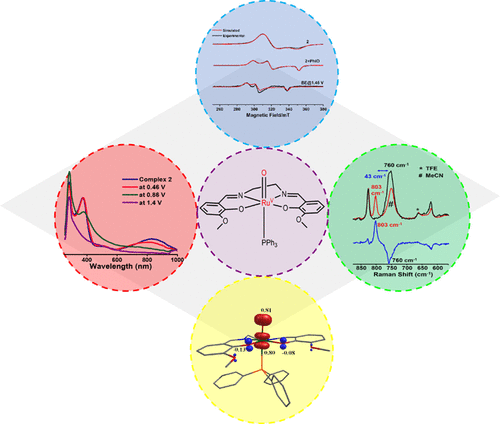
Beyond catalytic activity, our research strongly emphasizes a mechanistic understanding of these processes at the molecular level. We investigate how transition metal complexes activate small molecules, form reactive intermediates, and undergo multi-electron transfer steps. Particular attention is given to identifying and stabilizing transient species that govern catalytic pathways, as these intermediates provide critical insights into bond activation, electron transfer, and structure–property relationships.
To achieve this, we employ a diverse range of spectroscopic and analytical techniques. Electronic absorption (UV–Vis) spectroscopy is used to probe electronic structure and monitor reaction progress, while infrared (IR) and Raman spectroscopy provide detailed information on vibrational modes and ligand coordination. Electron paramagnetic resonance (EPR) spectroscopy plays a key role in detecting paramagnetic intermediates and understanding electronic environments in open-shell systems. Mass spectrometry enables the identification of reaction products and transient species, and X-ray diffraction (XRD) offers precise structural characterization of synthesized complexes.
By integrating synthetic chemistry with advanced spectroscopic techniques and mechanistic studies, our approach provides a comprehensive understanding of catalytic systems. This knowledge guides the rational design of next-generation catalysts with improved performance and stability. Overall, our work aims to contribute to the development of efficient and sustainable catalytic processes for energy conversion, environmental remediation, and the activation of small molecules.
Reference:
C–H Activation & Functionalization
Our research group is also actively working in the field of C–H activation/functionalization, a rapidly growing field that is transforming the way chemists design and build complex molecules. Interestingly, most pharmaceuticals and medicinally important compounds are largely composed of C–H bonds, which form the fundamental backbone of organic structures. However, despite being so common, C–H bonds are among the most chemically inert and difficult to modify selectively. This challenge has inspired the development of innovative strategies that can directly convert simple C–H bonds into valuable functional groups, enabling faster and more efficient synthesis of drug-like molecules.

Traditionally, chemists relied on pre-functionalized starting materials such as halides or organometallic reagents to construct complex structures. While effective, these approaches often require multiple synthetic steps, harsh reaction conditions, and generate significant chemical waste. In contrast, direct C–H functionalization provides a more step-economical and sustainable alternative by allowing chemists to “edit” molecules at a late stage, introducing new substituents without the need for lengthy preparation. This concept has become extremely important in medicinal chemistry, where rapid modification of lead molecules is essential for improving biological activity, selectivity, and pharmacological properties. At the heart of C–H functionalization lies the challenge of selectivity. Since most organic molecules contain many similar C–H bonds, activating one specific position without disturbing others is highly demanding. To overcome this, transition-metal catalysts such as palladium, rhodium, ruthenium, iridium, cobalt, nickel, and copper have been widely explored. These metals can activate C–H bonds through mechanistic pathways such as oxidative addition or concerted metalation–deprotonation (CMD), forming reactive metal–carbon intermediates. Once formed, these intermediates can undergo diverse transformations, enabling the formation of new C–C and C–heteroatom bonds, including olefination, arylation, amination, and halogenation.

In this context, our group is particularly interested in developing site-selective C–H functionalization of heteroarenes. We design and develop pincer-type isoquinoline-based directing templates that can precisely guide the catalyst toward the remote C5–H bond. This approach not only enables highly selective C5 functionalization but also provides a powerful platform for late-stage diversification of complex heteroarene scaffolds, offering new opportunities for drug discovery and molecular innovation.

Reference:
Photocatalysis
Light Driven CO₂ to Fuel Conversion via Molecular Catalysts
Development of homogeneous molecular photocatalytic systems for CO₂ reduction, where light harvesting and controlled electron transfer are coupled to convert carbon dioxide into value-added fuels. This approach enables efficient carbon capture and utilization through sunlight-driven chemical energy conversion.
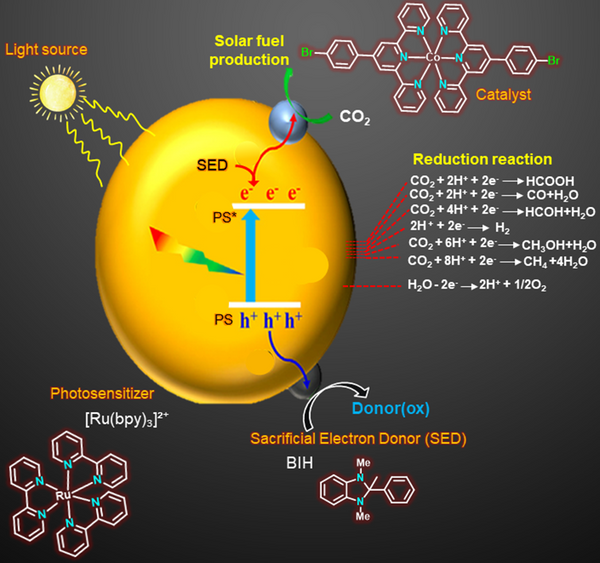
Light-driven homogeneous CO₂ reduction system
Our research focuses on the development of well-defined molecular catalyst systems for visible light driven CO₂ reduction. By integrating photosensitizers, molecular catalysts, and electron donors, efficient charge separation and transfer processes are achieved, enabling selective conversion of CO₂ into several value products. Through rational ligand design and tuning of metal centers, we aim to enhance catalytic efficiency, selectivity, stability and enhanced TON under operational conditions. This work aims to establish sustainable and tunable platforms for artificial photosynthesis, renewable fuel generation, and carbon-neutral technologies. Special emphasis is placed on understanding reaction mechanisms, electron transfer pathways, and catalyst structure property relationships to optimize performance.
Pyrovoltaics: Solar–Thermal Energy Harvesting
Study of nanocarbon framework (NCF)–based hybrid systems integrated with pyroelectric materials, where broadband light absorption and heat localization are coupled with temperature-dependent polarization to generate electrical output. This approach enables self-powered devices and sustainable energy technologies through efficient sunlight → heat → electricity conversion.
Solar-Thermal Pyroelectric Energy Harvesting
Our research focuses on the development of nanocarbon framework (NCF)–based hybrid systems integrated with pyroelectric materials for solar-driven energy harvesting. By exploiting the broadband light absorption and efficient heat localization of NCFs, we enable strong coupling with temperature-dependent polarization in pyroelectric materials, leading to direct electrical energy generation.
By designing hybrid systems that convert:
solar energy → thermal energy → electrical energy,
This work aims to establish efficient, lead-free, and scalable platforms for self-powered devices, thermal sensors, and sustainable energy technologies. Special emphasis is placed on understanding the structure–property relationship, interfacial heat transfer, and dipole dynamics to optimize device performance under real-world conditions.